|
This process is experimental and the keywords may be updated as the learning algorithm improves. These keywords were added by machine and not by the authors. These derivations are intended to give the reader a better understanding of the methods of calculating thermodynamic properties and thus of the limitations of the tables so produced. The purpose of this paper is to derive the functions necessary for the calculation of these properties. Considerable interest has been expressed about the derivation and application of these equations this interest generally results from the fact that standard texts on thermodynamics are, almost without exception, inadequate in the presentation of material on the calculation of thermodynamic properties. Where the hit occurred.Several recent publications from this laboratory on the calculation of thermodynamic properties of cryogenic fluids contain various relations for the determination of entropy, enthalpy, and internal energy. (energy) dissipates through air, when a book drops and hits the floor and theįloor gets warmer (molecules vibrate more rapidly) spreading out from the spot Heat (defined later) is random (disorganized) energy that is released orĪbsorbed, often as a result of some work being done, e.g., sound A heat reservoir (Figure 5.3) is a constant temperature heat source or sink. Work is organized energy, lifting a book, etc. Heat transfer from, or to, a heat reservoir. Potential Energy both of these can be forms of work and of heat energy Work done when gases are evolved or used up in the reaction. 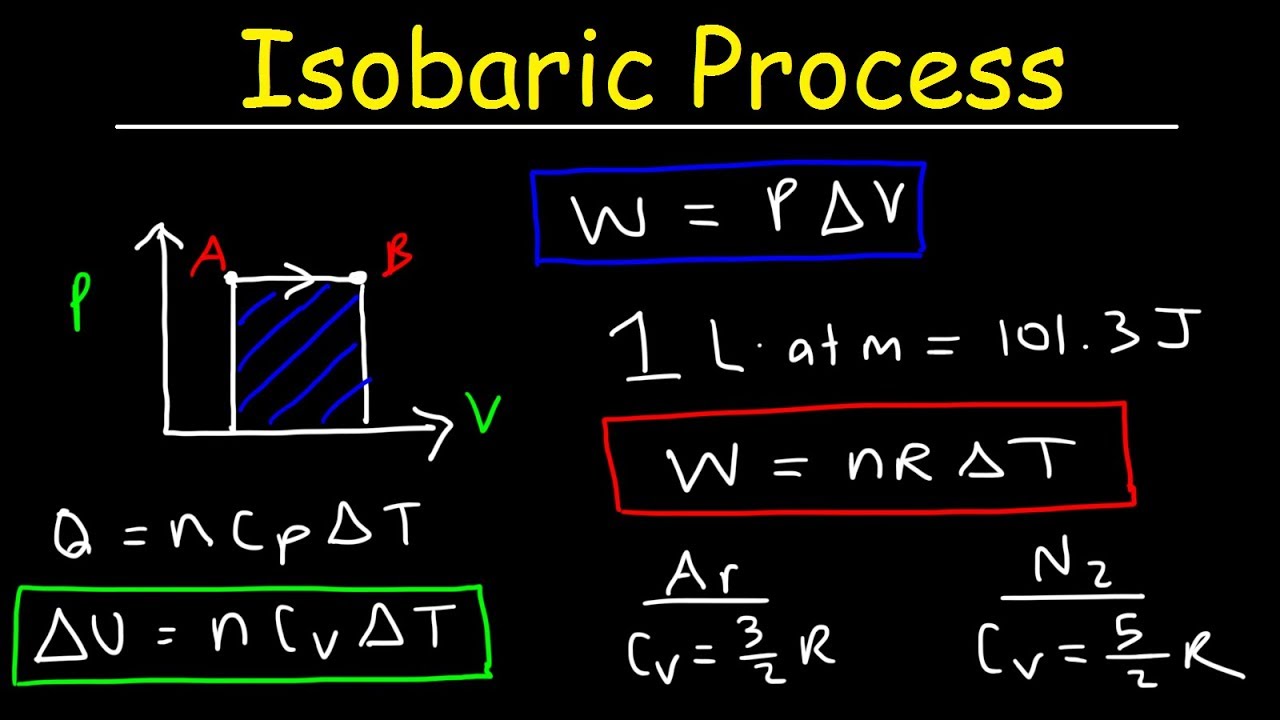
In chemical situations, we are normally interested only in the atmospheric It has used up energy so we can now write down a more complete definition for Hence, did the work of pushing back the atmosphere (positive value for Δ V), (Described later) Since the system increased in volume and The only thing left to decide is the signĬonvention. Hence, we can calculate the magnitude of the work The force needed to pushīack the atmosphere is actually P atm × A where the thermodynamic calculation of reaction equilibria. P atm and moves the piston a distance Δ x. Keywords: Alloys Enthalpy Entropy Heat capacity Inorganic compounds Metallurgy Thermodynamic. In this case, one pushes back against the atmospheric pressure Work can also be done by pushing back the atmosphere (piston in aĬylinder). This is a mechanical work and serves as the basis for our definition of PV Results (Calculation 1) Results (Calculation 2) Results (Calculation 3) Units (SI) Units (E) Tips. One of the most common definitions for work is when a force f displaces an object by a distance Δ xĭoes work w = f × Δ x. Calculate online thermodynamic and transport properties of water and steam, based on industrial (IAPWS-IF97) or scientific (IAPWS-95) formulation. Work and will use the symbol w to refer only to that type of work.Īll other types of work will be referred to using a prime on the symbol, In chemistry, we generally are only interested in PV Light-energy work and work of expansion or contraction of a gas ( PV There is mechanical work, electrical work, Energy The capacity to do work or to transfer heat Work Work is one means of transferring energy from our system to the Definitions: Thermodynamics The science of transformation of energy. Theseĭefinitions must be very tightly specified and understood in order to properlyįollow some of the more complex logic in later pages. Processes, we need to first develop a few terms that we will use. To begin the study of the transformation of energy in chemical (or other)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
